Regulatory authorities around the world are pursuing greater predictability, transparency, and harmonization. As a result, regulatory changes in Europe—such as the MDR/IVDR revision—and the implementation of unique device identification codes (UDI) in China are taking place simultaneously.
For professionals operating in the medical device sector, staying up to date makes all the difference. This article was created with that purpose: to keep you informed about the most relevant regulatory developments of the past few weeks.
Tip: Those who subscribe to our newsletter received all this content in advance. Don’t miss out—subscribe for free to receive the Sobel News Round Up.
European Union Signals Progress on MDR and IVDR Revisions
European Union authorities have expressed optimism regarding the proposed revisions to the Medical Device Regulation (MDR) and the In Vitro Diagnostic Regulation (IVDR).
Discussions focus on well-known implementation challenges since the regulations entered into force. Key issues include limited notified body capacity, challenging transitional timelines, and the significant administrative burden placed on manufacturers.
Although the proposals are still under evaluation, the positive signal suggests openness to adjustments based on real-world industry experience, without compromising device safety and performance requirements.
Do you manufacture medical devices and plan to access the European market? Then stay alert and reassess your regulatory strategies.
China Sets Final Deadline for Full UDI Compliance
Have you heard of the acronym UDI? It refers to unique device identification codes, a system designed to enable control and traceability of medical products.
UDI requirements are already being implemented in several countries. In China, authorities have announced that all medical devices must be fully compliant with UDI requirements by 2029.
This measure strengthens traceability throughout the product lifecycle, enhances post-market surveillance, and aligns China more closely with international regulatory practices.
In addition, the requirement increases the relevance of unique device identification codes as a central element for regulatory control, logistics, recalls, and safety monitoring.
Companies operating in—or planning to enter—the Chinese market must incorporate this requirement into their regulatory strategies as early as possible.
Tip: If your company is considering expanding operations and bringing products to China, one of the world’s largest markets, rely on Sobel!
We have specialists fully dedicated to regulatory operations in China, helping manufacturers navigate these processes with greater efficiency and confidence.
H2: Anvisa Launches Monitoring Panel for the 2026–2027 Regulatory Agenda
In Brazil, Anvisa has launched a public dashboard to monitor its 2026–2027 Regulatory Agenda.
The tool provides greater visibility into planned actions, timelines, and regulatory priorities. In practice, it strengthens transparency and enables more strategic planning for companies.
Another important development is Anvisa’s signing of the Rio de Janeiro Declaration, together with multiple Latin American regulatory authorities.
This milestone represents a commitment to regulatory convergence. The initiative seeks to enhance cooperation among agencies, streamline processes, and facilitate market access across the region.
Moreover, it is expected to promote alignment of technical and administrative requirements, including those related to unique device identification codes.
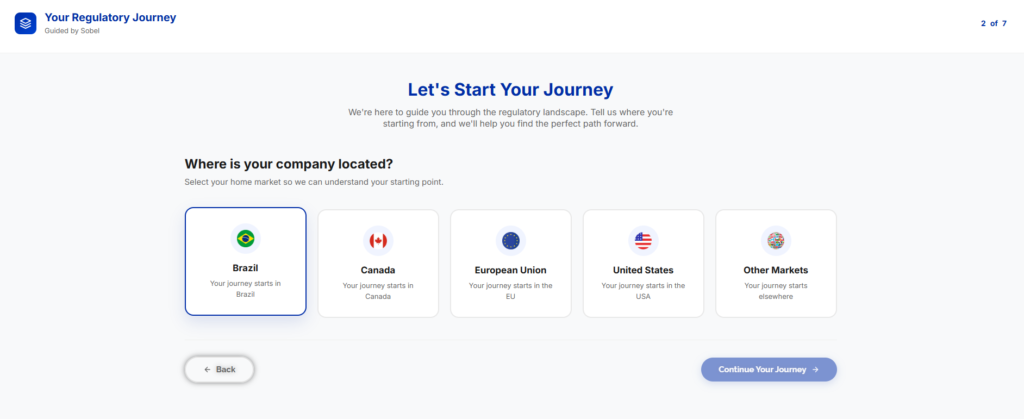
A New Tool on the Sobel Website
Do you want to regularize a medical device, cosmetic, or IVD but are unsure which service you need?
Sobel has the solution! We’ve launched a new tool on our website designed to guide you through your regulatory journey.
Here’s how it works: by clicking the chat button at the bottom of the website (or accessing the tool directly), you’ll answer a few questions.
At the end, the screen displays the services recommended for your case, along with the option to be contacted by our team of specialists.
It’s a practical and highly functional resource that will help you identify the best path for your regulatory process.
What Do These Changes Mean for Your Business?
As you’ve seen, many countries are making important moves in their regulatory frameworks.
Taken together, these initiatives reflect a global effort toward regulatory systems that are more predictable, integrated, and focused on patient safety.
That’s why staying informed is essential. It supports more secure strategic decision-making—whether for regulatory submissions or for implementing unique device identification codes across different markets.
Anticipating regulatory trends allows companies to adjust their roadmaps, optimize resources, and strengthen competitiveness in an increasingly regulated and interconnected environment.




