
Biological Evaluation, Clinical, and Toxicological: What’s the Difference?
In the development and approval process of medical devices, three
Welcome to our Publications page! Discover our latest articles, research papers, and reports, offering in-depth insights and expert analysis on various topics. Stay informed with cutting-edge research and industry trends. Explore our collection and keep up-to-date with the latest developments.

In the development and approval process of medical devices, three
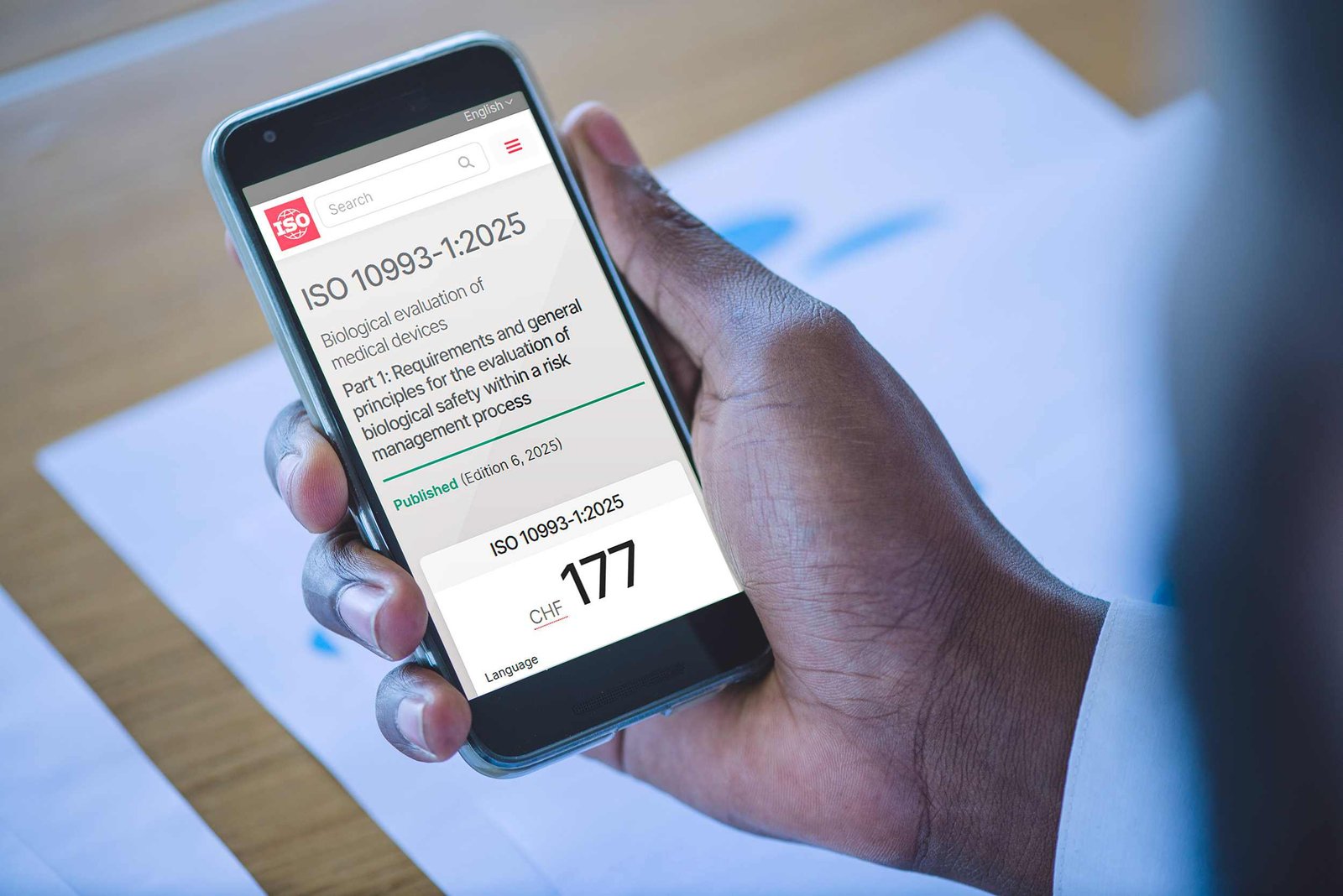
Learn how the updated ISO 10993-1 impacts biological risk assessments, gap analysis, documentation, and compliance for medical devices.

Learn how to conduct Biological Evaluations for Medical Devices using ISO 10993. Follow essential guidelines to ensure safety and compliance.
To provide the best experiences, we use technologies such as cookies to store and/or access device information. Consenting to these technologies allows us to process data such as browsing behavior or unique IDs on this site. Not consenting or withdrawing consent may negatively affect certain features and functions.